Who performed the gold foil experiment in 1911?
Rutherford
In 1911, Rutherford and coworkers Hans Geiger and Ernest Marsden initiated a series of groundbreaking experiments that would completely change the accepted model of the atom.
Who discovered the nucleus using gold foil?
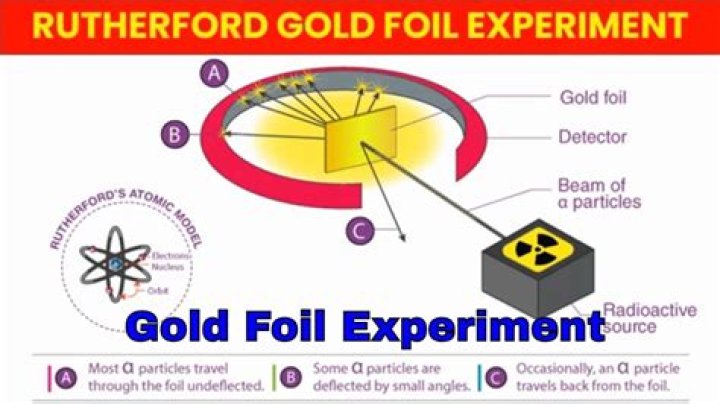
Rutherford overturned Thomson’s model in 1911 with his well-known gold foil experiment in which he demonstrated that the atom has a tiny and heavy nucleus. Rutherford designed an experiment to use the alpha particles emitted by a radioactive element as probes to the unseen world of atomic structure.
What was Rutherford’s experiment in 1911?
1911 John Ratcliffe and Ernest Rutherford (smoking) at the Cavendish Laboratory. In 1911, Rutherford discovered the atomic nucleus, using a “scattering” experiment, diagramed at right, that would become a classic technique of particle physics.
Which scientist performed an experiment with gold foil?
Physicist Ernest Rutherford established the nuclear theory of the atom with his gold-foil experiment.
What was Bohr’s model called?
planetary model
According to the Bohr model, often referred to as a planetary model, the electrons encircle the nucleus of the atom in specific allowable paths called orbits.
Who was involved in the gold foil experiment?
Gold Foil Experiment Who did the Gold Foil Experiment? The gold foil experiment was a pathbreaking work conducted by scientists Hans Geiger and Ernest Marsden under the supervision of Nobel laureate physicist Ernest Rutherford that led to the discovery of the proper structure of an atom.
What did Rutherford do with the gold foil?
Rutherford’s Gold Foil Experiment Animation Summary Rutherford demonstrated his experiment on bombarding thin gold foil with alpha particles contributed immensely to the atomic theory by proposing his nuclear atomic model.
Who was the first scientist to discover the nucleus?
The Geiger–Marsden experiment(s) (also called the Rutherford gold foil experiment) were a landmark series of experiments by which scientists discovered that every atom contains a nucleus where all of its positive charge and most of its mass are concentrated.
Why did Rutherford name his experiment the nucleus?
Rutherford named it the “nucleus” after experimenting with various gases. He also used materials other than gold for the foil, though the gold foil version gained the most popularity. He further went on to reject the plum pudding model and developed a new atomic structure called the planetary model.