Why is an alpha particle deflected in passing through gold foil?
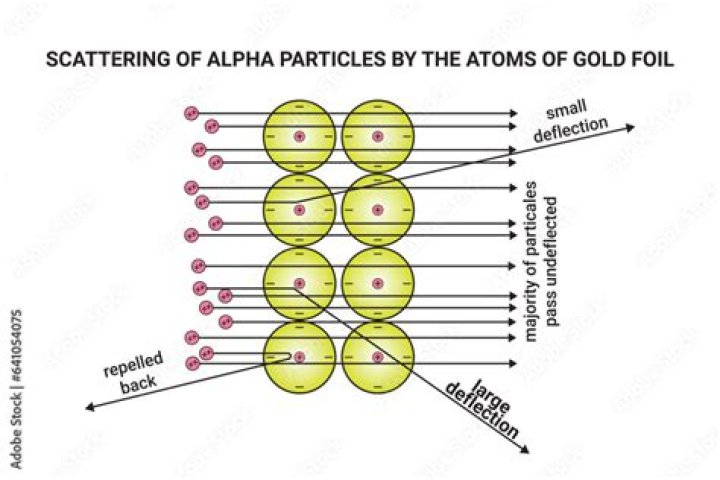
A small number of alpha particles were deflected by large angles (> 4°) as they passed through the foil. There is a concentration of positive charge in the atom. Like charges repel, so the positive alpha particles were being repelled by positive charges. Alpha particles were known to be much, much more dense than gold.
What happened to the alpha particles when they hit the gold foil Result 1?
A: The alpha particles would penetrate the gold foil. Alpha particles are positive, so they might be repelled by any areas of positive charge inside the gold atoms. Most of the alpha particles passed straight through the gold foil as though it wasn’t there. The particles seemed to be passing through empty space.
Does gold foil absorb alpha particles?
alpha rays pass though gold Even when a thin sheet of gold foil is placed in the path of the rays, most of them pass through it very easily.
Do alpha particles attract electrons?
As a result of its positive electric charge, an alpha particle is capable of attracting electrons from a relatively large distance. Pulling electrons away from many atoms, the alpha particle loses energy, slows down and finally stops.
What was the result of Rutherford’s gold foil experiment?
Rutherford’s gold foil experiment showed that the atom is mostly empty space with a tiny, dense, positively-charged nucleus. Based on these results, Rutherford proposed the nuclear model of the atom.
Can Alpha particles travel in vacuum?
Actually alpha particles travel great in a vacuum. Because the alpha is a large atom (~4 amu), and has a high charge (+2), it is highly ionizing as compared to other forms of radiation. In a vacuum it won’t have an electron cloud to interact with, so it’s smooth sailing.
Why are alpha particles deflected by gold foil?
This is due to the fact that like charges repel each other. As the positively charged alpha particle would fly through the foil it would come in proximity with the positively charge nucleus of the atom. This in turn either deflected the particle or adjusted its path.
Why do alpha particles have a + 2 charge?
Due to the positively charged nucleus of the gold atoms. Alpha particles are are positively charges particles that are made up of 2 protons, 2 neutrons and zero electrons. Due to the fact that protons have a +1 charge and neutrons hold no charge, this would give the particle a +2 charge over all.
Why did Rutherford think alpha particles would fly straight through foil?
Explanation: Alpha particles are are positively charges particles that are made up of 2 protons, 2 neutrons and zero electrons. Due to the fact that protons have a +1 charge and neutrons hold no charge, this would give the particle a +2 charge over all. Originally Rutherford thought that the particles would fly straight through the foil.
How does a nucleus cool down after emitting an alpha particle?
After emitting an alpha or beta particle, the nucleus will often still be too ‘hot’ and will lose energy in a similar way to how a hot gas cools down. A hot gas cools by emitting infrared radiation which is an electromagnetic wave.