Why is catalytic cracking important?
Catalytic cracking is now one of the most important processes practiced in petroleum refining as it allows the use of a much larger fraction of crude oil, converting relatively high molecular weight materials into high octane fuels.
What useful products are made from cracking?
Some of the smaller hydrocarbons formed by cracking are used as fuels (eg large chains are often cracked to form octane for petrol, which is in high demand), and the alkenes are used to make polymers in the manufacturing of plastics. Sometimes, hydrogen is also produced during cracking.
Why is thermal cracking used in industry?
Thermal cracking is currently used to “upgrade” very heavy fractions or to produce light fractions or distillates, burner fuel and/or petroleum coke. Two extremes of the thermal cracking in terms of the product range are represented by the high-temperature process called “steam cracking” or pyrolysis (ca.
Why would oil companies want to crack long alkanes?
Cracking the long chained alkanes produced from fractional distillation is necessary to produce ethene, propene and octane. When the process of hydrocarbon cracking is applied, long chain hydrocarbons become shorter ones. When crude oil is processed, it’s components have various boiling temperatures.
What are two conditions needed for cracking?
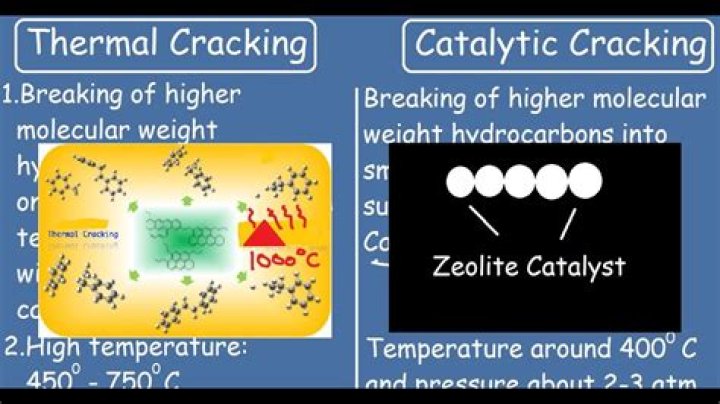
Thermal cracking uses harsh conditions like high temperature and high pressure. It breaks the alkanes into a high percentage of alkenes and comparatively few alkanes. Thermal cracking is done at about 1,000 degrees Celcius and 70 atm pressure.
What are the advantages of cracking?
Reasons for cracking Cracking is important for two main reasons: It helps to match the supply of fractions with the demand for them. It produces alkenes, which are useful as feedstock for the petrochemical industry.
What are the 2 types of cracking?
Cracking is primarily of two types – thermal cracking and catalytic cracking. Thermal cracking is further categorised into modern thermal cracking and steam cracking. On the other hand, sub-classifications of catalytic methods of cracking are hydrocracking and fluid catalytic cracking. 3.
What happens during cracking?
Cracking is a reaction in which larger saturated hydrocarbon molecules are broken down into smaller, more useful hydrocarbon molecules, some of which are unsaturated: the products of cracking include alkanes and alkenes , members of a different homologous series.
What is the process of cracking?
Cracking, in petroleum refining, the process by which heavy hydrocarbon molecules are broken up into lighter molecules by means of heat and usually pressure and sometimes catalysts. Cracking is the most important process for the commercial production of gasoline and diesel fuel.
What temperature is used for cracking?
Various methods can be used for cracking, eg catalytic cracking and steam cracking: Catalytic cracking uses a temperature of approximately 550°C and a catalyst known as a zeolite which contains aluminium oxide and silicon oxide. Steam cracking uses a higher temperature of over 800°C and no catalyst.
Why is cracking necessary to make crude oil?
Cracking converts long chain hydrocarbons into short chain hydrocarbons. Long-chain alkanes are broken down into alkanes and alkenes of shorter length. Crude oil contains a surplus long chains. Shorter chain hydrocarbons are in greater demand, e.g. petrol. Cracking also produces alkenes which are used in making polymers and ethanol.
Why is cracking important in the petrochemical industry?
Reasons for cracking. Cracking is important for two main reasons: It helps to match the supply of fractions with the demand for them. It produces alkenes, which are useful as feedstock for the petrochemical industry.
What kind of fuel is produced by cracking petroleum?
Cracking is the most important process for the commercial production of gasoline and diesel fuel. Cracking of petroleum yields light oils (corresponding to gasoline), middle-range oils used in diesel fuel, residual heavy oils, a solid carbonaceous product known as coke, and such gases as methane, ethane, ethylene, propane, propylene, and butylene.
Why is it important to avoid cracking in a refinery?
It is important to ensure that the feedstock does not crack to form carbon, which is normally formed at this temperature. This is avoided by passing the gaseous feedstock very quickly and at very low pressure through the pipes which run through the furnace.