Why is silver cyanide used in electroplating?
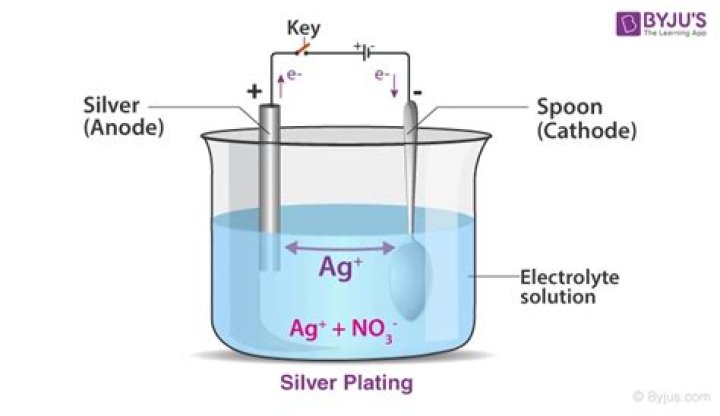
Sodium argentocyanide is a complex salt. Sodium silver cyanide is preferred to silver nitrate as an electrolyte for electroplating of an article with silver because the deposition of silver ion is slow and smooth and the coating is smooth.
What chemical is used for gold plating?
Potassium gold cyanide is the most important gold plating chemical. Almost all the gold used for contacts in the electronic industry is derived from potassium gold cyanide.
What is the difference between potassium cyanide and sodium cyanide?
The cyanide used in the L-pill was potassium cyanide but the properties of sodium cyanide are nearly identical. An inorganic and very innocent looking white solid with deadly properties, sodium cyanide (NaCN) can be fatal at amounts as little as 5% of a teaspoon.
What is gold plating solution?
Gold plating uses an alkaline gold plating solution to deposit gold onto a metal substrate by electrolysis. Following the old tradition, the gold plating solution used cyanide compounds, which are poisonous and must be dispensed in a very restrictive manner.
Does cyanide dissolve gold?
A sodium cyanide solution is commonly used to leach gold from ore. The cyanide dissolves the gold from the ore into the solution as it trickles through the heap.
Is gold plating toxic?
Although gold is not a threat to the environment, the cyanide used in the gold plating bath is very toxic and listed as one of 17 “high- priority” chemicals by the U.S. EPA. Therefore, it is environmentally desirable to find an alternative plating bath that does not contain cyanide, nickel or cobalt.
What is the best type of gold plating?
If you’re looking for the highest-quality, demi-fine jewelry, a vermeil collection with thick plating in 18k gold would be a good choice. If you don’t mind a lower base metal, gold filled pieces are probably your best bet when it comes to long-lasting and durable jewelry.
Where is cyanide found?
Cyanides can be produced by certain bacteria, fungi and algae. Cyanides are also found in cigarette smoke, in vehicle exhaust, and in foods such as spinach, bamboo shoots, almonds, lima beans, fruit pits and tapioca.
Why are gold and silver alloys soluble in cyanide?
Inasmuch as gold-silver alloys of this composition are almost as soluble in cyanide as gold is, it is probable that some other factor has affected the solubility of the electrum particles. Possibly some other alloying element has influenced their solubility in cyanide.
How are simple cyanides different from complex cyanide?
Cyanides are classified as simple or complex cyanides: Simple cyanides are represented by the general form A(CN)ₓ where A is an alkali (sodium, potassium or ammonium) or a metal (such as copper or zinc), and x (the valence of A) is the number of CN groups. -Simple alkali cyanides dissociate easily in aqueous solutions.
Is there a difference between nitrile and cyanide?
We generally use the terms cyanide and nitrile interchangeably because both these terms refer to the C≡N group or the cyano group.
When did they start separating gold with cyanide?
Several patents were issued in the same year. By suspending the crushed ore in a cyanide solution, a separation of up to 96 percent pure gold was achieved. The process was first used on the Rand in 1890 and, despite operational imperfections, led to a boom of investment as larger gold mines were opened up.