Why silver nitrate is used in electroplating?
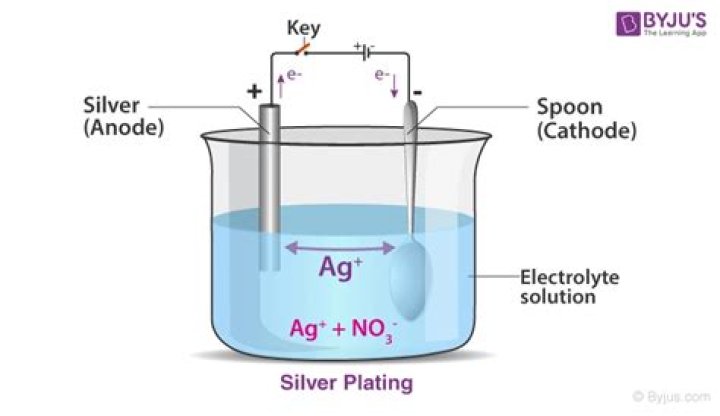
In the illustration shown below it is a metal salt of silver, silver nitrate (AgNO3), that has been dissolved to create a solution of silver ions (Ag+) and nitrate ions (NO3-). Electroplating is a widely used technique because it can plate a variety of metals onto a variety of substrates to produce smooth coatings.
Why is silver nitrate not a good electrolyte?
A solution of AgNO3 is a good electrolyte but is not used for electroplating an article with silver because it undergoes very fast redox process and electroplating done through this process is not uniform.
Will silver nitrate react with silver?
Silver nitrate can be prepared by reacting silver, such as silver bullion or silver foil, with nitric acid, resulting in silver nitrate, water, and oxides of nitrogen. Reaction byproducts depend upon the concentration of nitric acid used.
Why is silver nitrate bad?
► Exposure to Silver Nitrate can cause headache, dizziness, nausea and vomiting. to transport Oxygen, causing headache, fatigue, dizziness, and a blue color to the skin and lips (methemoglobinemia). Exposure to very high levels can cause trouble breathing, collapse and even death.
Is silver nitrate bad conductor of electricity?
(c) Both solid silver (Ag) and molten Ag are good conductors of electricity. However, solid silver nitrate, AgNO3, is a good conductor only when melted or dissolved in pure water; as a solid, it is a poor conductor.
Is silver nitrate an electrolyte?
The compound silver nitrate is a strong electrolyte.
Is silver nitrate a good electrolyte?
Is silver nitrate a weak acid?
Is silver nitrate acidic or basic? – Quora. According to Lewis theory of acid-base, cation of metals have empty orbitals and acts as an acid. silver ion has empty orbital thus solution must be acidic. nitrate ion is conjugate base of a strong acid thus its basicity is weak.
What chemicals does silver react with?
Silver reacts readily with sulfur or hydrogen sulfide (H2S) to produce silver sulfide (Ag2S), a dark-colored compound familiar as the tarnish on silver coins and other objects. Silver sulfide also forms silver whiskers when silver electrical contacts are used in an atmosphere rich in hydrogen sulfide.
Which is better for electroplating silver or silver nitrate?
In the electroplating of an article with silver, the electrolyte sodium argento-cynide solution is preferred over silver nitrate solution. If silver nitrate solution is used directly instead of double cyanide of silver and sodium, the deposition of silver will be very fast and hence not very smooth and uniform.
Can You electroplate a mirror with silver nitrate?
You can not practically electroplate with silver nitrate. It is used for plating mirrors with an old process using sugar and formaldehyde. Go back to the main page, find the search engine and type in silver plating. About a year ago a person added the proper name for this process.
Can a silver nitrate solution be used instead of sodium cyanide?
If silver nitrate solution is used directly instead of double cyanide of silver and sodium, the deposition of silver will be very fast and hence not very smooth and uniform. Is there an error in this question or solution?
What happens to silver nitrate when it is heated?
When silver reacts with hot and concentrated nitric acid, it emits nitrogen dioxide. Silver Nitrate is a salt, which is colorless or of a white crystalline form. When this is exposed to light or any organic material, this becomes black in color. Silver Nitrate when heated decomposes and forms, Silver, Nitrogen dioxide and Oxygen.